Protection is not as good as mRNA vaccine. What are the advantages of inactivated vaccine?
On December 30, 2020, Beijing Institute of Biological Products Co., Ltd. quietly published a short announcement in official website. According to the interim analysis results of Covid-19 inactivated vaccine phase III clinical trial, the safety of Covid-19 inactivated vaccine of China Bio-Beijing Company of Sinopharm Group was good after inoculation.After two doses of immunization, the positive conversion rate of neutralizing antibody was 99.2%, and the protective power of vaccine against diseases caused by COVID-19 infection (Covid-19) was 79.34%.
Prior to this, Pfizer and Moderna from the United States also released the research results of phase III clinical trials of their own mRNA vaccines, and the protective power was over 90%. In this way, our vaccines seem to have a little poor protective effect. But be careful,This is not because our technology is not good, but more because of the congenital difference between mRNA vaccine and inactivated vaccine.
Immune protection induced by inactivated vaccine is relatively weak.
Inactivated vaccine is a vaccine made by inactivating virus by physical or chemical methods. Compared with live virus particles, the immunogenicity of inactivated virus is relatively weak, and adjuvant is often needed.
In 2013, a study entitled "Characterization of immune responses induced by activated, live attended and DNA vacuums against Japanese encephalitis virus in mice" was confirmed by Japanese encephalitis virus.Compared with artificial attenuated live vaccine and DNA vaccine, the immune response induced by inactivated vaccine is relatively weak.
Figure 1. Heading of the article
The researchers divided the mice into four groups, injected with inactivated vaccine (INV), live attenuated vaccine (LAV), DNA vaccine (pCAG-JME) and control, and then detected the antibody level in mice. At the end of the experiment, the total antibody titer induced by inactivated vaccine was significantly lower than that of attenuated live vaccine group and DNA vaccine group.
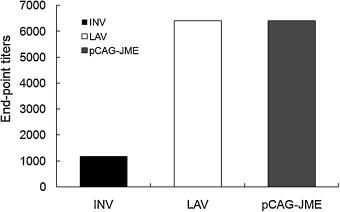
Fig. 2 Antibody level pairs induced by three vaccines
The researchers also detected the levels of different subtypes of IgG induced in the serum of mice, and found that there were differences among the three vaccines. The inactivated vaccine mainly induced IgG type 1 antibody, while the attenuated live vaccine mainly induced IgG type 2a antibody, and the antibody levels of DNA vaccine were both high.
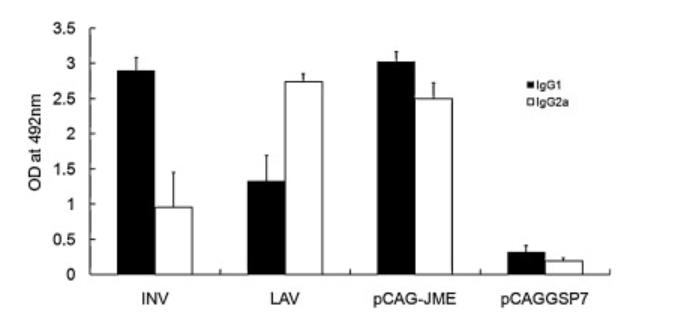
Fig. 3 Different IgG subtypes induced by three vaccines
The researchers also compared the levels of cytokines induced by vaccine and the levels of dendritic cells and Treg in detail, and there were some differences. Anyway,There are some differences in immune response induced by inactivated vaccine, attenuated live vaccine and nucleic acid vaccine. It is not surprising that the protective effect of inactivated vaccine is not as good as that of RNA vaccine.
Inactivated vaccine has the best safety.
Because the clinical data of phase III vaccine of Beijing Institute of Biological Products have not been published in any authoritative journals,Therefore, the safety of inactivated vaccine BBIBP-CorV in China, mRNA vaccine BNT162b2 in Pfizer and mRNA vaccine mRNA-1273 in Moderna Company are examined from the published phase I/II clinical trials of vaccines.Someone has made statistics on the safety data of phase I/II clinical trials of various COVID-19 vaccines, and all the data are shown in the following figure. After the first dose of mRNA-1273 vaccine was injected, the incidence of systemic grade I adverse reactions was 53.3%, grade II adverse reactions were 13.3%, local grade I adverse reactions were 73.3%, and grade II adverse reactions were 13.3%. After injecting the second dose, the incidence of systemic first-class adverse reactions was 20%, and the incidence of second-class adverse reactions was 80%. The incidence of local first-class adverse reactions was 66.7%, and the incidence of second-class adverse reactions was 26.7%. No adverse reactions above grade 3 were found locally or systemically.
However, after injection of Pfizer’s BNT162b2, the incidence of local or systemic adverse reactions was 41.7%, and the incidence of adverse events above grade 3 was 8.3%. For people aged 65-85, the incidence of local or systemic adverse reactions is 25%, and the incidence of adverse reactions above grade 3 is 8.3%.
The results of phase I clinical trial of domestic inactivated vaccine BBIBP-CorV in China show that the incidence of first-grade local or systemic adverse reactions is 33% for people aged 18-59, and that of second-grade adverse reactions is 13%. For people over 60, the incidence of adverse reactions is 29%, and there are no adverse events above grade 3. In the results of Phase II clinical trials, the incidence of Grade I adverse events was 15%, and the incidence of Grade II adverse events was 2%.
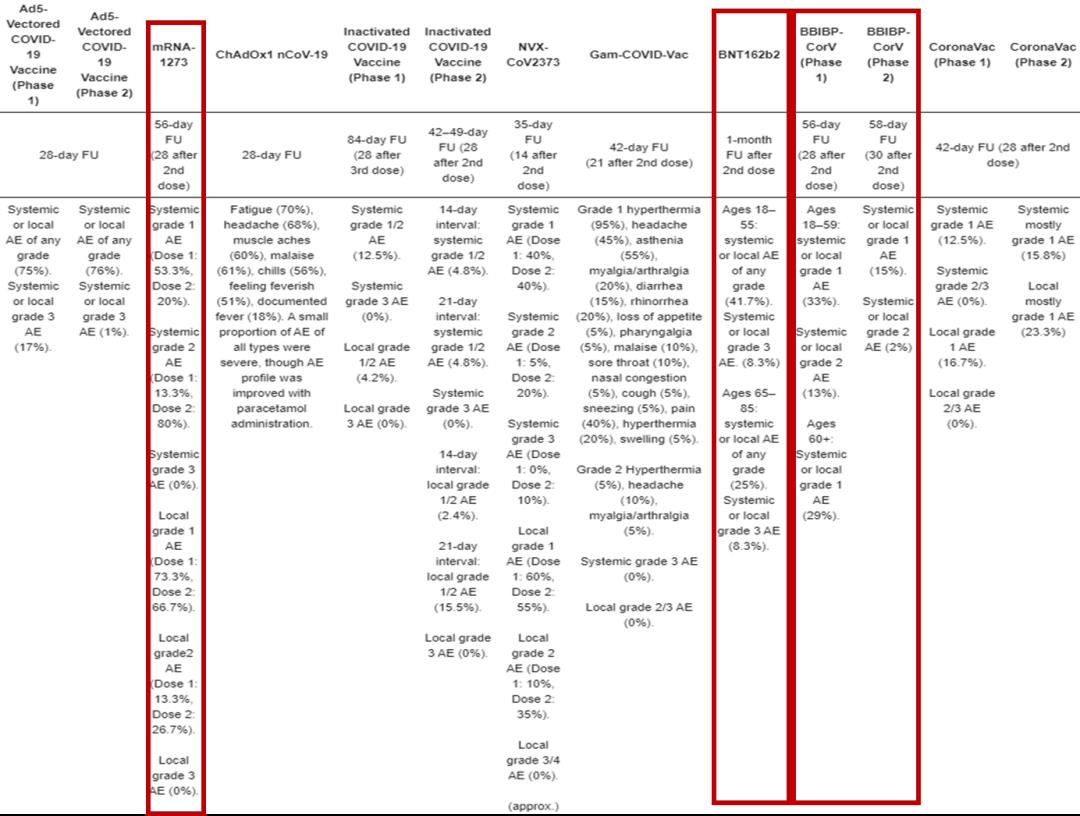
Fig. 4 Adverse reactions of various vaccines.
Judging from these data, among the three popular vaccines at present, the safety of inactivated vaccine in China is absolutely the best, not to mention the recent allergic storm of Pfizer vaccine, which has caused rare allergic reactions in dozens of people.
Domestic vaccines with 79% protection and no serious adverse reactions are absolutely safe and effective.Because China’s epidemic prevention and control work is very well done, we have to overcome many difficulties when conducting clinical trials of vaccines. After all, the epidemic situation in COVID-19 is well controlled and there is no condition for clinical trials, which is one of the reasons why the progress of phase III clinical trials lags behind others. We should see that even if our protection efficiency does not exceed 90% in the United States, inactivated vaccine obviously has advantages, such as it is safer and easier to store and transport. Although our protective effect is not as good as the two vaccines in the United States, it has reached the standard of vaccine approval, and the protective effect far exceeds 50%, which is trustworthy.
